The Toxicological Process
Definition
Toxicology is the study of poisons.
Poisons are chemical/physical agents that produce adverse responses in biological organisms.
What is there that is not poison?
All things are poison and nothing without poison. Solely, the dose determines that a thing is not a poison”
Paracelsus (1493-1541).
The Toxicological Paradigm
Toxicology is the study of poisons.
Poisons are chemical/physical agents that produce adverse responses in biological organisms.
What is there that is not poison?
All things are poison and nothing without poison. Solely, the dose determines that a thing is not a poison”
Paracelsus (1493-1541).
The Toxicological Paradigm
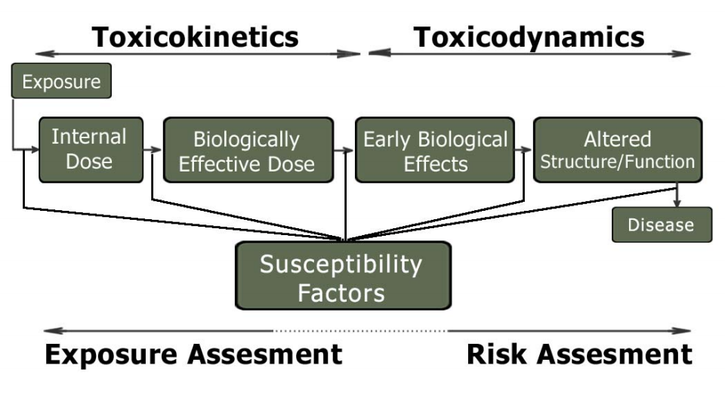
Toxicokinetics
Toxicokinetics is the quantitation of the time course of toxicants in the body during the processes of absorption, distribution, biotransformation, and excretion or clearance of toxicants.The end result of these toxicokinetic processes is a biologically effective dose of the toxicant.
Toxicodynamics
Toxicodynamics refers to the molecular, biochemical, and physiological effects of toxicants or their metabolites in biological systems.
These effects are result of the interaction of the biologically effective dose of the ultimate (active) form of the toxicant with a molecular target.
Molecular Targets Concept
The toxic action of a chemical is a consequence of the physical/chemical interaction of the active form of that chemical with a molecular target within the living organism.
Examples of Molecular Targets
Proteins
Arylhydrocarbon(Ah) receptor—Dioxin
Hemoglobin—CO
Lipids—Carbon tetrachloride
DNA—Aflatoxin
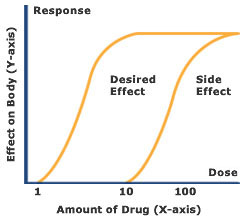
Dose-Response Concept
The magnitude of the toxic effect will be a function of the concentration of altered molecular targets, which in turn is related to the concentration of the active form of the toxicant( biologically effective dose) at the site where the molecular targets are located.
Toxicokinetics is the quantitation of the time course of toxicants in the body during the processes of absorption, distribution, biotransformation, and excretion or clearance of toxicants.The end result of these toxicokinetic processes is a biologically effective dose of the toxicant.
Toxicodynamics
Toxicodynamics refers to the molecular, biochemical, and physiological effects of toxicants or their metabolites in biological systems.
These effects are result of the interaction of the biologically effective dose of the ultimate (active) form of the toxicant with a molecular target.
Molecular Targets Concept
The toxic action of a chemical is a consequence of the physical/chemical interaction of the active form of that chemical with a molecular target within the living organism.
Examples of Molecular Targets
Proteins
Arylhydrocarbon(Ah) receptor—Dioxin
Hemoglobin—CO
Lipids—Carbon tetrachloride
DNA—Aflatoxin
Dose-Response Concept
The magnitude of the toxic effect will be a function of the concentration of altered molecular targets, which in turn is related to the concentration of the active form of the toxicant( biologically effective dose) at the site where the molecular targets are located.
Section B
Transport Process Mechanisms
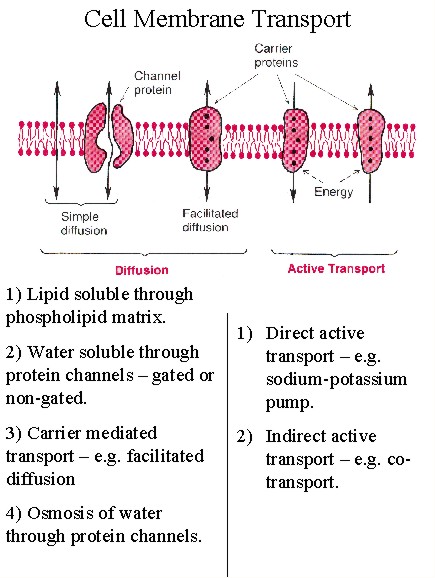
Membrane Transport of Xenobiotics
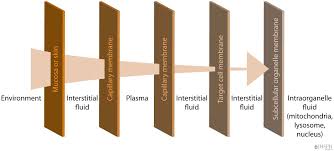
The absorption, distribution, and excretion of xenobiotics involves passing through various cell and organ membranes. This occurs through various transport mechanisms.
Definition Of Xenobiotics
Pharmacologically or toxicologically active substance not endogenously produced and therefore foreign to an organism.
Xenobiotics: Transport Mechanisms
Factors affecting membrane transport of chemicals:
•Molecular weight/shape
•Charge
•Lipid solubility
•Membrane composition
Membrane thickness
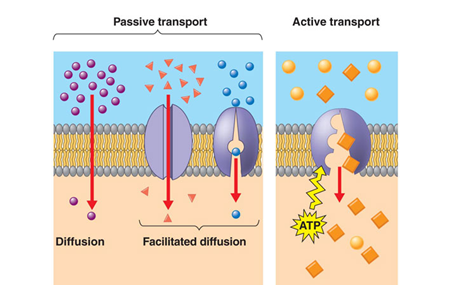
Types of Transport
•Simple diffusion
•Facilitated diffusion
•Active transport
•Pinocytosis
•Filtration
Transport By Plasma Membrane
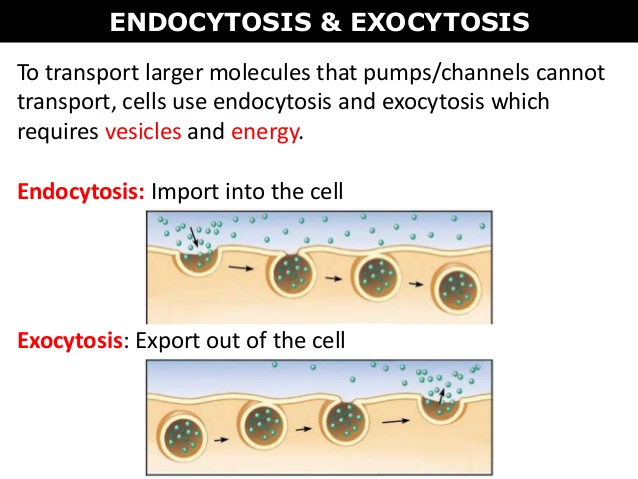
Exo- and Endocytosis
Exocytosis - “cell vomiting“ is a release of larger molecules by the protrusion of a cellular membrane, under delivery of energy and Ca2+ ions
4Endocytosis –“cell eating“ is an uptake of molecules by a cellular membrane, e.g. ingestion of bacteria by leukocytes (phagocytosis). It needs a delivery of energy, too.
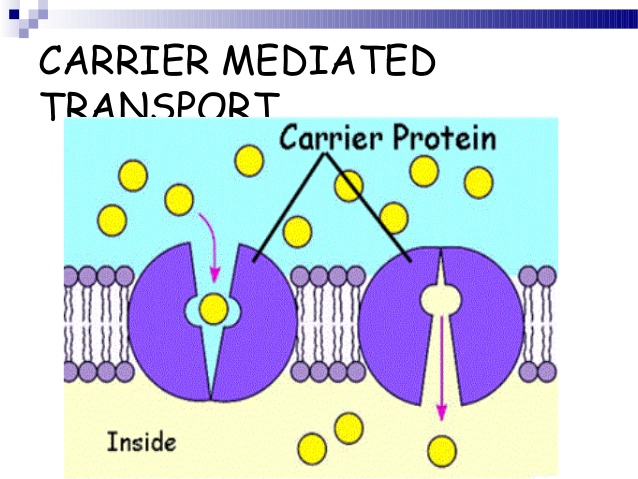
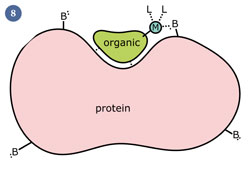
BOTH PASSIVE MEDIATED and ACTIVE MEDIATED TRANSPORT INVOLVE the USE of CARRIER PROTEINS
Carrier-Mediated Transport
Carrier-Mediated Transport
Characteristics of Passive Mediated Transport
•Transport proceeds in the direction of the electrochemical potential (concentration) gradient.
•Structural specificity (specific inhibitors)
•No energy requirements
•Inherently symmetrical transport
Characteristics of Active Mediated Transport
•Transport can proceed against an electrochemical potential (concentration) gradient
•The process is saturable at high concentration gradients
•Structural specificity
•Requires cellular energy
Asymmetrical transport
Filtration
•Transport of solutes as a consequence of bulk flow of fluid (aqueous) phase
•Glomerulus of kidney is a good example of site where filtration occurs
Section C
Toxicokinetics
Systemic Kinetics: Outline
•Physiological basis of toxicokinetics
•Biliary excretion route for foreign compounds
•Barriers
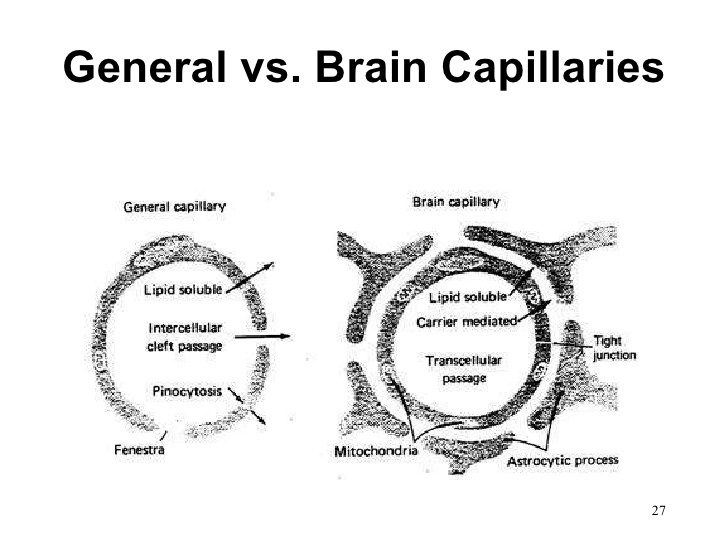
•Major difference between a general (non-neural) and brain capillary
•Excretion pathways
•Transport of solutes as a consequence of bulk flow of fluid (aqueous) phase
•Glomerulus of kidney is a good example of site where filtration occurs
Section C
Toxicokinetics
Systemic Kinetics: Outline
•Physiological basis of toxicokinetics
•Biliary excretion route for foreign compounds
•Barriers
•Major difference between a general (non-neural) and brain capillary
•Excretion pathways
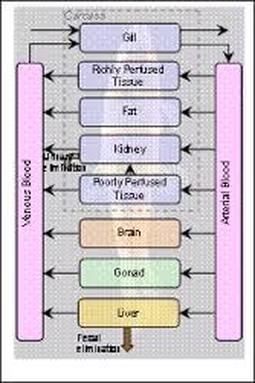
Systemic Kinetics: Barriers
- Blood-brain barrier
- Placenta
- Blood-testicular barrier
Major Difference between a General (Non-Neural) and a Brain Capillary
- Blood-brain barrier
- Placenta
- Blood-testicular barrier
Major Difference between a General (Non-Neural) and a Brain Capillary
Excretion Pathways
•Respiratory excretion ( Mucocilliary clearance).
•Gastrointestinal excretion (Biliary excretion, Entero-hepatic circulation).
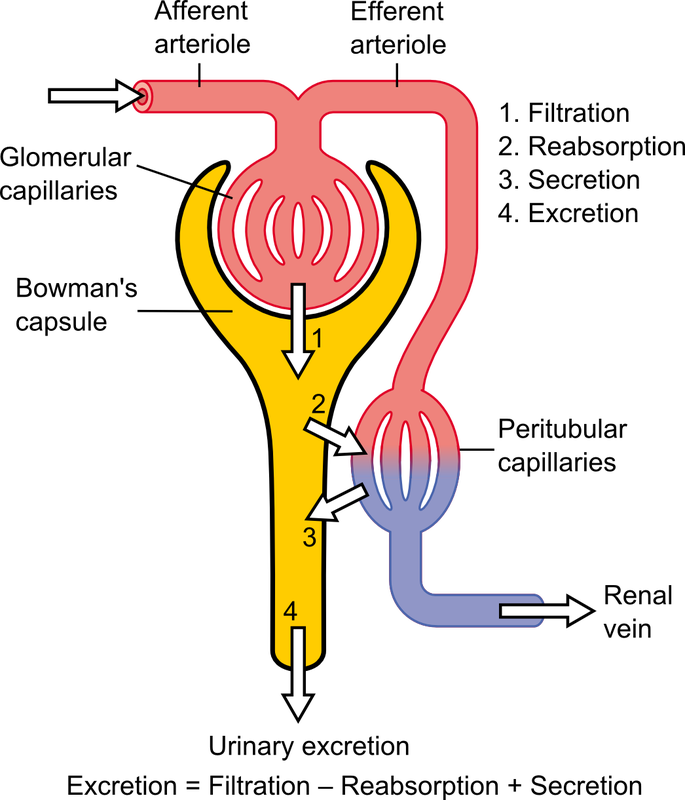
•Urinary excretion ( Glomerular filtration, Trans-tubular secretion).
Renal Excretion of Chemicals
•Respiratory excretion ( Mucocilliary clearance).
•Gastrointestinal excretion (Biliary excretion, Entero-hepatic circulation).
•Urinary excretion ( Glomerular filtration, Trans-tubular secretion).
Renal Excretion of Chemicals
Other Routes of Excretion
•Milk
•Sweat
•Hair
•Nails
•Saliva
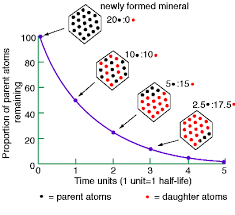
Biological Half Life
The biological half-life(T1/2) is the time required for some measure of the amount of a chemical in the body (for example, body burden, tissue concentration) to decrease to 1/2 its value at the beginning of the observational interval.
•Milk
•Sweat
•Hair
•Nails
•Saliva
Biological Half Life
The biological half-life(T1/2) is the time required for some measure of the amount of a chemical in the body (for example, body burden, tissue concentration) to decrease to 1/2 its value at the beginning of the observational interval.
Section D
Toxicokinetics and PBK Modeling
PBPK Modeling
Physiologically Based Pharmacokinetic (Toxicokinetic)
PurposeTo mathematically model how a substance is absorbed, distributed, and metabolized in the body to reduce uncertainties in determining the estimated dose.
The PBPK models attempt to mimic disposition of drugs in the body by using measured values of such parameters as:
-tissue- and blood-volumes
-cardiac output
-metabolic clearance rates (e.g. knowledge on the kinetic rates of P450 enzymes)
-absorption rate (e.g. skin or gastric emptying)
-various partition coefficients (tissue:blood or blood:air).
Generally, these parameters fall in either of three different categories:
•Biochemical
•Physiological
•partition coefficients
A simple PBPK model may include blood, brain, kidney, and liver. Each compartment is specialised and limited by perfusion and diffusion.
In other cases, each organ may also be further divided into sub-compartments, such as the stomach content of the stomach, or the free drug vs. protein-bound drug.
The use of PBPK also allows for more complex models that may target subgroups of the population, such as pregnant women or children.
Toxicokinetics and PBK Modeling
PBPK Modeling
Physiologically Based Pharmacokinetic (Toxicokinetic)
PurposeTo mathematically model how a substance is absorbed, distributed, and metabolized in the body to reduce uncertainties in determining the estimated dose.
The PBPK models attempt to mimic disposition of drugs in the body by using measured values of such parameters as:
-tissue- and blood-volumes
-cardiac output
-metabolic clearance rates (e.g. knowledge on the kinetic rates of P450 enzymes)
-absorption rate (e.g. skin or gastric emptying)
-various partition coefficients (tissue:blood or blood:air).
Generally, these parameters fall in either of three different categories:
•Biochemical
•Physiological
•partition coefficients
A simple PBPK model may include blood, brain, kidney, and liver. Each compartment is specialised and limited by perfusion and diffusion.
In other cases, each organ may also be further divided into sub-compartments, such as the stomach content of the stomach, or the free drug vs. protein-bound drug.
The use of PBPK also allows for more complex models that may target subgroups of the population, such as pregnant women or children.
Vinyl Chloride Metabolism
Toxic Chemicals
The toxicity of a chemical refers to its ability to:
Damage an organ system (kidneys, liver), disrupt a biochemical process (e.g., the blood-forming process) or disturb an enzyme system at some site remote from the site of contact.
Dose Time Relationship
The most important factor is the dose-time relationship. The dose-time relationship forms the basis for distinguishing between two types of toxicity: acute toxicity and chronic toxicity.
The acute toxicity
The acute toxicity of a chemical refers to its ability to inflict systemic damage as a result (in most cases) of a one-time exposure to relative large amounts of the chemical. In most cases, the exposure is sudden and results in an emergency situation.
Chronic toxicity
Chronic toxicity refers to a chemical's ability to inflict systemic damage as a result of repeated exposures, over a prolonged time period, to relatively low levels of the chemical.
Some chemicals are extremely toxic and are known primarily as acute toxins (hydrogen cyanide); some are known primarily as chronic toxins (lead). Other chemicals, such as some of the chlorinated solvents, can cause either acute or chronic effects.
The toxic effects from chemical exposure depend on the severity of the exposures. Greater exposure and repeated exposure generally lead to more severe effects.
Methods Of Chemicals Exposure
Exposure to toxic chemicals can occur by:
- Inhalation
- Dermal absorption
- Ingestion
- Injection
Inhalation and dermal absorption are the most common methods of chemical exposure in the workplace.
Types Of Toxic Chemicals
•Toxicants
•Carcinogens
•Reproductive Toxins
•Sensitizers
•Irritants
Acute Toxins
Acute toxins can cause severe injury or death as a result of short-term, high-level exposure. Examples of acute toxins: Hydrogen cyanide, Hydrogen sulfide, Nitrogen dioxide, Ricin, Organophosphate pesticides, Arsenic.
Chronic Toxins
Chronic toxins cause severe injury after repeated exposure.
Examples of chronic toxins include the following:
•Mercury
•Lead
•Formaldehyde
Carcinogens
Carcinogens are materials that can cause cancer in humans or animals.
Examples of known carcinogens include the following:
•Asbestos
•Benzene
•Tobacco smoke
•Chromium, hexavalent
Aflatoxins
Reproductive Toxins
Reproductive toxins are chemicals that can produce adverse effects in parents and developing embryos. Chemicals including:
Sensitizers
Repeated exposures may result in severe allergic reactions.
Examples of sensitizers include the following:
•Isocyanates
•Nickel salts
•Beryllium compounds
•Formaldehyde
•Diazomethane
Irritants
Irritants cause reversible inflammation or irritation to the eyes, respiratory tract, skin, and mucous membranes through long-term exposure or high concentration exposure. Examples of irritants include:
•Ammonia
•Formaldehyde
•Halogens
•Sulfur dioxide
.Poison ivy , Phosgene
•Heavy metals,
•Some aromatic solvents (benzene, toluene, xylenes, etc.),
•Some therapeutic drugs, ionizing radiation, alcohol consumption, cigarette smoking and illicit drugs.
The toxicity of a chemical refers to its ability to:
Damage an organ system (kidneys, liver), disrupt a biochemical process (e.g., the blood-forming process) or disturb an enzyme system at some site remote from the site of contact.
Dose Time Relationship
The most important factor is the dose-time relationship. The dose-time relationship forms the basis for distinguishing between two types of toxicity: acute toxicity and chronic toxicity.
The acute toxicity
The acute toxicity of a chemical refers to its ability to inflict systemic damage as a result (in most cases) of a one-time exposure to relative large amounts of the chemical. In most cases, the exposure is sudden and results in an emergency situation.
Chronic toxicity
Chronic toxicity refers to a chemical's ability to inflict systemic damage as a result of repeated exposures, over a prolonged time period, to relatively low levels of the chemical.
Some chemicals are extremely toxic and are known primarily as acute toxins (hydrogen cyanide); some are known primarily as chronic toxins (lead). Other chemicals, such as some of the chlorinated solvents, can cause either acute or chronic effects.
The toxic effects from chemical exposure depend on the severity of the exposures. Greater exposure and repeated exposure generally lead to more severe effects.
Methods Of Chemicals Exposure
Exposure to toxic chemicals can occur by:
- Inhalation
- Dermal absorption
- Ingestion
- Injection
Inhalation and dermal absorption are the most common methods of chemical exposure in the workplace.
Types Of Toxic Chemicals
•Toxicants
•Carcinogens
•Reproductive Toxins
•Sensitizers
•Irritants
Acute Toxins
Acute toxins can cause severe injury or death as a result of short-term, high-level exposure. Examples of acute toxins: Hydrogen cyanide, Hydrogen sulfide, Nitrogen dioxide, Ricin, Organophosphate pesticides, Arsenic.
Chronic Toxins
Chronic toxins cause severe injury after repeated exposure.
Examples of chronic toxins include the following:
•Mercury
•Lead
•Formaldehyde
Carcinogens
Carcinogens are materials that can cause cancer in humans or animals.
Examples of known carcinogens include the following:
•Asbestos
•Benzene
•Tobacco smoke
•Chromium, hexavalent
Aflatoxins
Reproductive Toxins
Reproductive toxins are chemicals that can produce adverse effects in parents and developing embryos. Chemicals including:
Sensitizers
Repeated exposures may result in severe allergic reactions.
Examples of sensitizers include the following:
•Isocyanates
•Nickel salts
•Beryllium compounds
•Formaldehyde
•Diazomethane
Irritants
Irritants cause reversible inflammation or irritation to the eyes, respiratory tract, skin, and mucous membranes through long-term exposure or high concentration exposure. Examples of irritants include:
•Ammonia
•Formaldehyde
•Halogens
•Sulfur dioxide
.Poison ivy , Phosgene
•Heavy metals,
•Some aromatic solvents (benzene, toluene, xylenes, etc.),
•Some therapeutic drugs, ionizing radiation, alcohol consumption, cigarette smoking and illicit drugs.