Haematology
Zoo. 506
Part II
Anesthesia and Blood Collection
Rodent Anesthesia
Definitions:
COMMONLY USED ANESTHETICS By:
Zoo. 506
Part II
Anesthesia and Blood Collection
Rodent Anesthesia
Definitions:
- Loss of consciousness in addition to loss of sensation; ideally includes hypnosis, hyporeflexia, analgesia, and muscle relaxation; can be produced using a single drug (i.e. Isoflurane) or a combination of drugs (i.e. Ketamine and Xylazine).
COMMONLY USED ANESTHETICS By:
- INHALANT AGENTS
- Gases or Vapours
- INJECTABLE ANESTHETICS

INHALANT AGENTS
- Isoflurane and Halothane.
- Although some of the following agents are still used in clinical practice and in research.
- aliflurane, chloroethane, chloroform, cyclopropane, diethyl ether, enflurane, ethylene, halothane, methoxyflurane, methoxypropane, roflurane, teflurane,trichloroethylene and vinyl ether.
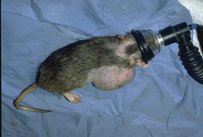
Gases or vapours
- Gases or vapours which produce general anaesthesia by inhalation include nitrous oxide, cyclopropane and xenon.
INJECTABLE ANESTHETICS
Ketamine
Ketamine
- Ketamine is a dissociative anesthetic used in a wide variety of species
- barbiturates still have their place in the research animal laboratory.
- Avertin has been the standard anesthetic in much mouse transgenic work (125-250 mg/kg i.p.). It can produce short-term (15-20 minutes) surgical anesthesia with good muscle relaxation and moderate respiratory depression.
Intravenous administration
Intravenous simply means "within vein". Compared with other routes of administration, the intravenous route is the fastest way to deliver fluids and medications throughout the body.
Intravenous simply means "within vein". Compared with other routes of administration, the intravenous route is the fastest way to deliver fluids and medications throughout the body.
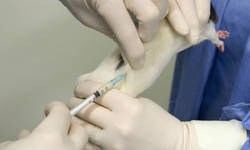
Subcutaneous injection
A subcutaneous injection is administered as a bolus into the subcutis, the layer of skin directly below the dermis and epidermis, collectively referred to as the cutis. Subcutaneous injections are highly effective in administering vaccines and medications such as insulin, morphine, diacetylmorphine and goserelin.
A subcutaneous injection is administered as a bolus into the subcutis, the layer of skin directly below the dermis and epidermis, collectively referred to as the cutis. Subcutaneous injections are highly effective in administering vaccines and medications such as insulin, morphine, diacetylmorphine and goserelin.
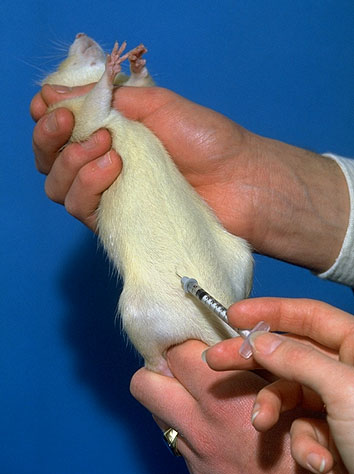
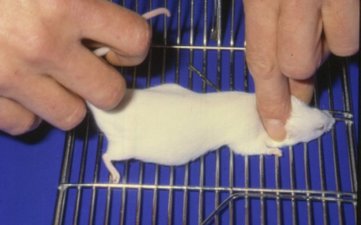
Intraperitoneal injection
Intraperitoneal injection or IP injection is the injection of a substance into the peritoneum (body cavity). IP injection is more often applied to animals than to humans.
Intraperitoneal injection or IP injection is the injection of a substance into the peritoneum (body cavity). IP injection is more often applied to animals than to humans.

Gastric Administration
Material can be administered orally using a number of different techniques, although gavage using a stomach tube is the most widely used.
Material can be administered orally using a number of different techniques, although gavage using a stomach tube is the most widely used.

SPECIES-SPECIFIC CONSIDERATIONS
Mice
- In general, smaller animals have higher metabolic rates and frequently require higher doses of anesthetics at more frequent intervals to achieve the desired effect.
Mice
- Isofluran is recommended as the first choice anesthetic in mice. It should be delivered at a known percentage (1-3% for maintenance; up to 5% for induction) in oxygen from a precision vaporizer.
-
Rats
- Rat anesthesia considerations are similar to those described for the mouse, though some doses vary.In rats, ketamine combinations with xylazine (75-100mg/kg + 16 mg/kg i.p.) are more likely to provide adequate surgical anesthesia than in mice and so may not require supplemental isoflurane.
Hamsters
Hamster anesthesia is similar to rat and mouse anesthesia, though some anesthetic doses differ.
Hamster anesthesia is similar to rat and mouse anesthesia, though some anesthetic doses differ.

Guinea Pigs
- Intraperitoneal (IP) administration works well, if the large cecum can be avoided.
- Guinea pigs may be anesthetized by face mask with gas anesthetics.
Blood Collection Of Animals
Part III
Blood Collection of Rat
- Tail blood collection.
- Blood sample collection from rat orbital sinus
- Blood sample collection through cardiac puncture in rat.
- How to obtain blood samples from the facial vein of a mouse.
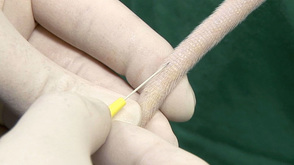
Tail blood collection
- Tail vein sampling is suitable for all strains and for competent individuals, is quick and simple to perform. However, this technique requires the rats to be warmed in order to dilate the blood vessel prior to taking the sample.
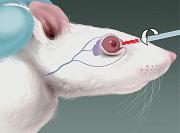
Blood sample collection from rat orbital sinus
Blood is collected from the venous sinus. The mouse is restrained, the neck gently scruffed and the eye made to bulge. A capillary tube/pipette is inserted medially, laterally or dorsally. Blood is allowed to flow by capillary action into the capillary tube. The sample obtained is a mixture of venous blood and tissue fluid, and is not representative of venous blood.
Blood is collected from the venous sinus. The mouse is restrained, the neck gently scruffed and the eye made to bulge. A capillary tube/pipette is inserted medially, laterally or dorsally. Blood is allowed to flow by capillary action into the capillary tube. The sample obtained is a mixture of venous blood and tissue fluid, and is not representative of venous blood.
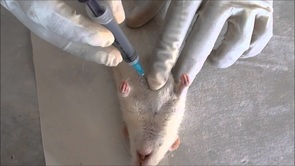
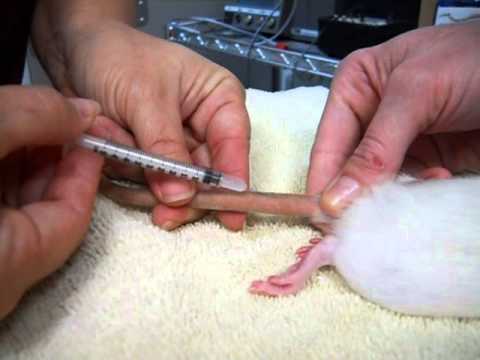
Blood sample collection through cardiac puncture in rat
Cardiac puncture is a suitable technique to obtain a single, large, good quality sample from a euthanised rat or a rat under deep terminal anaesthesia if coagulation parameters, a separate arterial or venous sample or cardiac histology are not required. It is appropriate for all strains.
Cardiac puncture is a suitable technique to obtain a single, large, good quality sample from a euthanised rat or a rat under deep terminal anaesthesia if coagulation parameters, a separate arterial or venous sample or cardiac histology are not required. It is appropriate for all strains.
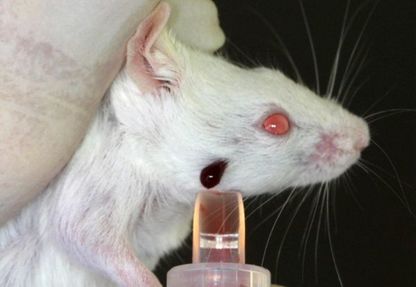
Blood Samples from the facial vein of a mouse
Blood collection from the submandibular vein is the preferred blood collection technique for mice. This method allows for maximum allowable sample volume with minimal trauma to the animal.
Blood collection from the submandibular vein is the preferred blood collection technique for mice. This method allows for maximum allowable sample volume with minimal trauma to the animal.
Collection of blood by decapitation
Trunk blood is collected from the site where the animal is decapitated. Training for decapitation should be undertaken on dead animals. Trunk blood is collected from the site where the animal is decapitated. Training for decapitation should be undertaken on dead animals.
Trunk blood is collected from the site where the animal is decapitated. Training for decapitation should be undertaken on dead animals. Trunk blood is collected from the site where the animal is decapitated. Training for decapitation should be undertaken on dead animals.
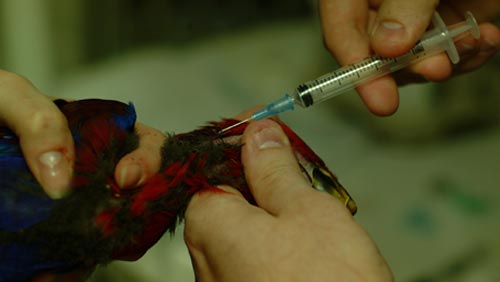
Blood Sampling Of Bird
- The large vein under the wing (brachial vein)
- Place the bird on a table, setting it on its side.
- Lift up the wing with one hand and part the feathers along the wing. Water can be used to help keep the feathers separated.
- Place the needle at a slight angle, bevel up, against the vein on the underside of the wing. (The bevel is the side of the needle with the angle and the hole.) Insert the needle into the vein and slowly withdraw blood.
- The vein on the side of the outstretched neck (jugular vein)
- Place the bird on a table, setting it on its side.
- Stretch out the neck with one hand and part the feathers along the neck. The right jugular vein is usually larger.
- Place the needle at a slight angle, bevel up, against the vein.
- Puncture the vein and slowly withdraw blood.
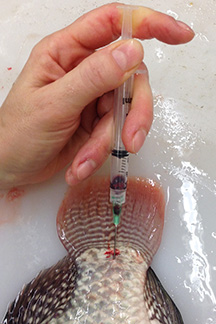
Blood Sampling of Fish
Blood samples are typically taken from the caudal vein, which sits directly under the vertebral column of the fish.
Blood samples are typically taken from the caudal vein, which sits directly under the vertebral column of the fish.
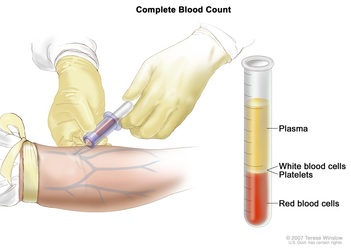
Blood Sampling Of Human
Blood samples should be taken from the vein in the antecubital fossa. Before blood collection, the subject should remove tight clothes that might constrict the upper arm. During blood collection, the arm should rest on a pillow or other supportive prop.
Blood samples should be taken from the vein in the antecubital fossa. Before blood collection, the subject should remove tight clothes that might constrict the upper arm. During blood collection, the arm should rest on a pillow or other supportive prop.
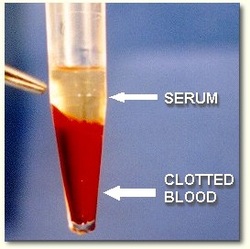
Serum Preparation
- Collect whole blood in a covered test tube.
- Allow the blood to clot by leaving it at room temperature.
- This usually takes 15-30 minutes.
- Remove the clot by centrifuging at 1,000-2,000 x g for 10 minutes in a refrigerated centrifuge.
- stored, and transported at –20°C or lower.
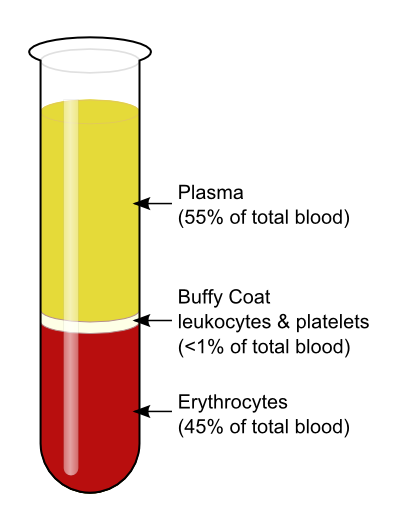
Plasma Preparation
- Collect whole blood into commercially available anticoagulant-treated tubes e.g., EDTA-treated or citrate-treated.
- Cells are removed from plasma by centrifugation for 10 minutes at 1,000-2,000 x g using a refrigerated centrifuge.
- Centrifugation for 15 minutes at 2,000 x g depletes platelets in the plasma sample.
- The resulting supernatant is designated plasma.
References
- Dennis O'Neil (1999). "Blood Components". Palomar College. Archived from the original on June 5, 2013.
- Tuskegee University (2013): "Chapter 9 Blood". tuskegee.edu. Archived from the original on December 28, 2013.
- Maton, Anthea; Jean Hopkins; Charles William McLaughlin; Susan Johnson; Maryanna Quon Warner; David
- LaHart; Jill D. Wright (1993): Human Biology and Health. Englewood Cliffs, New Jersey, USA: Prentice Hall. ISBN 0-13-981176-1.
- "19th WHO Model List of Essential Medicines (April 2015)" (PDF). WHO. April 2015. Retrieved May 10, 2015.
- Masoud M, Sarig G, Brenner B, Jacob G (June 2008):"Orthostatic hypercoagulability: a novel physiological mechanism to activate the coagulation system". Hypertension. 51 (6): 1545–51.
- Starr, Douglas P. (2000): Blood: An Epic History of Medicine and Commerce. New York: Quill. ISBN 0-688-17649-
- Gale ,R.P. (1994): stem cell transplants. New York, Cambridge University Press.
- Yoburn BC, Morales R, Inturrisi CE.(1984): Chronic vascular catheterization in the rat: Comparison of three techniques. Physiol Behav.;33:89–94.