Cellular Metabolism

Cellular metabolism is the sum total of all the biochemical reactions taking place within a cell. It includes all the reactions involved in degrading food molecules, in synthesizing macromolecules needed by the cell, and in generating small precursor molecules, such as some amino acids , for cellular needs. It also includes all reactions involving electron transfers (oxidation reduction, or redox, reactions). Metabolism takes place in sequences of biochemical reactions called pathways.
METABOLISM
The term metabolism refers to all of thebiochemical reactions by which complex molecules taken into an organism are broken down to produce energy and by which energy is used to build up new molecules.
All metabolic reactions fall into one of two general categories:
1.Anabolic Reactions
2.Catabolic Reactions
Anabolism
It is a metabolic process wherbyenergy is consumed to synthesize amino group substances acids transformationinto protein.
Catabolism
It is a metabolic process occurring in living cells by which complex molecules are broken down to produce small and usable amount of energy to sustain life.
Energy and Food
Foods can be oxidized with O2 in the cell with releasing energy. The same foods can also burned with pure oxygen outside body in fire, releasing same amount of energy ( in the form of heat). Foods can be oxidized with O2 in the cell with releasing energy. The same foods can also burned with pure oxygen outside body in fire, releasing same amount of energy ( in the form of heat).
Free Energy
The amount of energy liberated by complete oxidation of food is called the free energy of food (∆F). Free energy (∆F) is expressed in terms of calories/mol of food substance.
ATP and Metabolism
The amount of free energy in the third and second phosphate of high energy bonds per mole of ATP is 8000 cal. under standard condition.
Released and Reform Energy
Released Energy
ATP – 8000 cal ADP – 8000 cal AMP
Reform Energy
ATP + 8000 cal ADP + 8000 cal AMP
+ PO4 + PO4
Basal Metabolic Rate (BMR)
It is the mimimal caloric requirement needed to sustain life in a resting state i.e. amount of energy of body would burn if you slept all day (24 hours).
Physiological Factors That Affect BMR
Age: In youth, BMR is higher, age slows BMR.
Height: Tall, thin people have higher BMR.
Growth: Children and pregnant have higher BMR.
Body composition: The more lean tissue, the higher BMR, more fat tissue, the lower BMR.
Fever: Fever can rise BMR.
Stress: Stress hormone can rise the BMR.
Envivonmental temperture: Both the heat and cold raise BMR.
Fasting / starvation : Fasting /Starvation lower BMR.
Malnutrition: It lowers the BMR.
Thyroxin: The thyriod hormone is key BMR regulator, more thyroxin, the higher BMR.
Sympathetic stimulation: Liberation of norepinephrine and epinephrine increase the metabolic rate. These hormones directly affect cells to cause glycogenolysis.
Calculation Of BMR
BMR is calculated either by: General calculation or Harris-Benedict calculation
General Calculation
BMR=Body weight in lbs x10 kcal/lb
Ex.
Ahmed weighs 150 lbs.
BMR = 150 x 10 kcal/lb = 1500 kcals.
Harris-Benedict Calculation
Males: 66+(13.7x W) + (5 x H) - (6.8 x A)
Females: 655+ (9.6 x W)+(1.7 x H) - (4.7 x A)
W = weight in kg (weight in lb/2.2 lb/ kg.
H = height in cm (height in inches x 2.54 cm/in).
A = age in years.
Ex.
Ahmed weighs 150 lbs, stands 56”, and is 21 years old.
150lbs/2.2lb/kg = 68kg. and stands is 56''=56 inches x 2.54cm=168c
BMR=66+(13.7x 68)+ (5x 168)–(6.8 x 21)
BMR = 66 + 932 + 840 - 143 = 1695 kcals/day.
Maintaining the Basal Metabolic Rate (metabolism) accounts for 60–75% of daily energy expenditure in most people, while physical activity accounts for only 10–15%.
Energy Expenditure
Energy expenditure shows approximate energy during performance at various physical activities. i.e. how many hours you spent walkings, standing, running, exercising, sleeping and so on.
Ex.
Let’s say that Hassan spent 10 hours sitting, 3 hours walking, 1 hrs standing, 3 hrs studying, 1 hrs of running at 7.5mph, and 6 hrs of sleeping. This is Hassan’s activty record for that day. Now we can use the following charts to calculate how much energy burned per day.
Charts
1.1Kcals/minute burned during sleep i.e 1.1 ×360
minutes (6 hours) = 369 kcals.
1.7 kcals /minute burned during studying i.e 1.7× 130 minute (3 hours) = 306 Kcals.
2.5 kcals /mintues burned during walking at 3.5 mph i.e. 2.5× 180 (3 hours) = 936 Kcals.
14.1 Kcals/minutes burned during running at 7.5 mph i.e. 14.1 × 60 (1 hour) = 846 Kcals.l2.5 /kcals /mintues burned during standing i.e. 2.5× 60 (1 hours) = 150 Kcals.
1.5 kcals /mintues burned during sitting i.e. 1.5× 60 minute (10 hours) = 900 Kcals.
Energy expenditure = 396 + 306 + 936 + 846 + 150 + 900 = 3534 Kcals during this day.
Hassan needs to consume this much in his diet to maintain his body weight. If he wanted to gain weight, he would have to eat more.
If he wanted to lose weight, he would have to expend more energy with exercise and partially cut his food intake.
Respiratory Quotient (RQ)
The respiratory quotient (RQ) measures the ratio of the volume of CO2 (Vc) produced by an organism to the volume of O2 consumed (Vo).
The equation is: RQ= V CO2/V O2
RQ for Carbohydrate
The metabolic reaction of a carbohydrate is described
by glucose oxidation as the following equation:
C6H12O6 (glucose) + 6O2 = 6CO2 + 6H2O R.Q =6/6=1.
RQ for Fat
In fat oxidation; the tripalmitin is oxidized as the following equation:
2C51H98O6+145 O2=102 CO2+98 H2O.
The oxidation of fat or protein the R.Q. will be less than 1(0.7 in fat and 0.8 in protein).
Body Temperature
-The normal body temperature ranged from 36.5 oC to 37.5 oC
-Excessive heat is produced in exercise (38.5 to 40 oC).
-When it was measured by rectum, the value are » 0.6 oC greater than the oral temperatures.
FACTORS THAT AFFECT THE BODY TEMPERATURE
(1) Diurnal Rythm: It is fluctuated during 24 hours from lowest at morning and higher inafternoon.
(2) Age: The regulation of temp. In children is less from that ofthe adult.
(3)Site of Measured Temperature: It is differed in mouth compared by rectal site.
(4)Sex: It differs in female from male e.g estrous cycle in female rises 04-07oC at time of ovulation due to thermogenic effect of progesterone hormone.
(5)Emotions: It rises during emotional excitement due to muscular tone & activation of sympathetic system.
(6)Activity: It is increased during muscular excersise (2-3oC).
(7)Environmental: Temperature: It increased during high environmental temperature and vice versa. (8)Endocrinal Factors: It is high in hyperthyroidism and low in hypothyroidism.
Heat Balance
When the rate of heat production is equal to the rate of loss, the body is said to be in heat balance.
i.e. Rate of Heat gain = Rate of Heat Loss
Heat Gain
Factors leading to heat gain are:
Basal metabolic processes: It produces heat.
Food intake: It produces heat
Muscular activity: It increases heat production.
Endocrinal Factors: Catecholamines and thyrods hormones.
Brown fat: It is found in infants but absent in adult human. It contains large numbers of special mitochondria and supplied with sympathetic nerve fibres.
Heat Loss
The heat loss include:
-Radiation (60%) ,
-Conduction (3%),
-Evaporation (22%) and
-Convection (15%).
Radiation
Loss of heat by radiation (60%) means loss in the form of infrared heat rays (electromagnetic wave, its length of 5-20 micrometers).
Evaporation
-Water evaporates from the skin and lungs at rate of about 450 to 600 ml/day. This causes continual heat loss at rate of 12 to 16 calories /hour.
Conduction
-Small heat quantities (3%) are normally lost from the body by conduction from the body surface to solid objects.
Convection
It is the removal of heat from the body by convection air currents (15%).
Thermoregulatory System
It Consists of :
1.Receptors (thermoreceptors)
2.Afferent neurons
3.Control centre in hypothalamus
4.Efferent neurons
5.Effector organs
Sweeting
Large quantities of sweat are secreted into the skin surface by sweat glands to provide rapid evaporative cooling of the over heated body.
-In cold weather, the rate of sweat production is zero.
-In very hot the maximum rate of sweat production is from 1 L/hour to 3 L/hour.
-Sweat contains some other chemicals like ammonia , urea, salt and sugar. Urea and ammonia are waste products from breaking down protein when digesting. Sweat comes out from the skin pores.
-Through the logic of heat transfer the water ( sweat ) comes in contact with the air and gets transformed to water vapor thus cooling ourselves.
Sweeting and Its Regulation
-The preoptic area in the anterior part of hypothalamus is stimulated to excite sweating.
-The nerve impulses from this area are transmitted to a spinal cord and then through sympathetic outflow to the skin.
-Glands can also be stimulated by epinephrine or norepinephrine.
Hyperthermia
-Hyperthermia is overheating.
-In people suffer heat-related illness, the body normally cools itself by sweating.
-When sweating isn't enough,a body temperature rises rapidly.
-Very high body temperatures may damage the brain or other organs.
Factors limiting control of Temperature
Several conditions that can limit the ability to regulate temperature include:
-High humidity.
-Old age.
-Obesity.
-Fever.
-Dehydration.
-Heart disease.
-Poor circulation.
-Sunburn.
-Drug and alcohol use.
Carbohydrate metabolism
-Carbohydrate catabolism is the breakdown of carbohydrates into monosaccharide.
-Monosaccharide (glucose, fructose and galactose) are absorbed through portal vein into liver and carried to the cells of the body by circulation.
Mobilization of Carbohydrate Reserves In Insect
Glycogen is mobilized for use by other tissues, mostly in the form of trehalose.
The utilization of glycogen depends on the activity of glycogen phosphorylase, which provides glucosyl residues for trehalose
synthesis. Fat body phosphorylase activity increases during larval development. An increase in activity prior to pupation is linked to the need for energy and glucose for chitin synthesis. Likewise, pupal-adult development is marked by high phosphorylase activity.Different insects exhibit different patterns of phosphorylase activity during the pupal period.
In some insects the increase in phosphorylaseactivity is observed at the beginning of the period, whereas in others phosphorylase activity increases toward the end of the pupal stage .
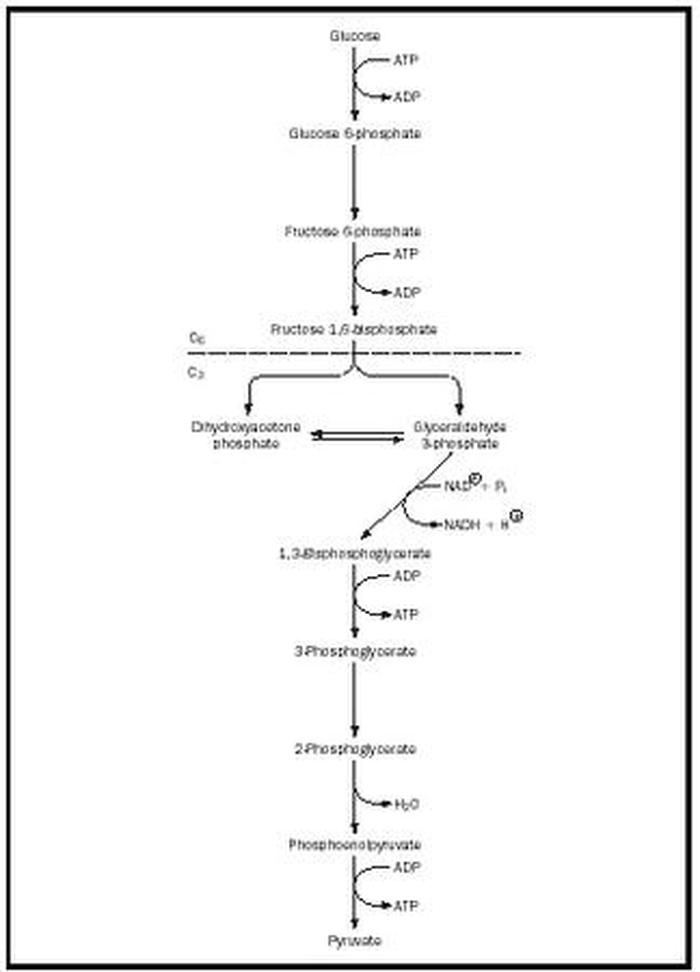
Glycolysis
It is known that complete oxidation of 1 mol of glucose release 686.000 calories of energy and 8000 calories are required to form 1 mol of ATP. Cells contatin different enzymes that cause the glucose molecule to split a litte a time in many successive steps with energy released in small packets to form one ATP at a time.
Phosphorylation Of Monosaccharides
Glucose + ATP Glucokinase Glucose -6-P
Fructose + ATP Fructokinase Fructose -6- P
Galactose + ATP Galactokinase Galactose -6-P
It is known that complete oxidation of 1 mol of glucose release 686.000 calories of energy and 8000 calories are required to form 1 mol of ATP. Cells contatin different enzymes that cause the glucose molecule to split a litte a time in many successive steps with energy released in small packets to form one ATP at a time.
Phosphorylation Of Monosaccharides
Glucose + ATP Glucokinase Glucose -6-P
Fructose + ATP Fructokinase Fructose -6- P
Galactose + ATP Galactokinase Galactose -6-P
Glycolysis
Glycogenesis
It is the process of glycogen formation. Lactic acid, glycerol, pyruvic acid and some deaminated amino acid can converted into glucose and thereby into glycogen.
Glycogenesis
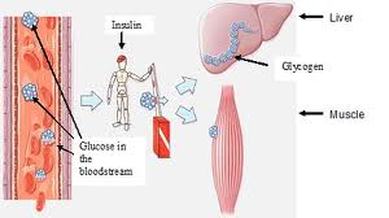
It is the process of glycogen formation. Lactic acid, glycerol, pyruvic acid and some deaminated amino acid can converted into glucose and thereby into glycogen.
Glycogenolysis
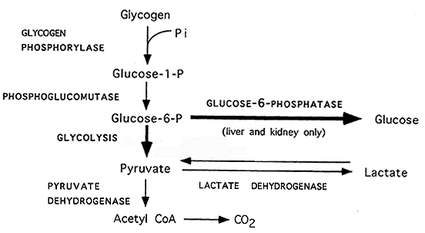
It is the process of glycogen breakdown. Glycogenolysis
does not occure by reversal chemical reactions as that manner to form glycogen, but each glucose molecule on branch of glycogen is split away by phosphorylase enzyme i.e. several enzymes split glycogen.
does not occure by reversal chemical reactions as that manner to form glycogen, but each glucose molecule on branch of glycogen is split away by phosphorylase enzyme i.e. several enzymes split glycogen.
Gluconeogenesis
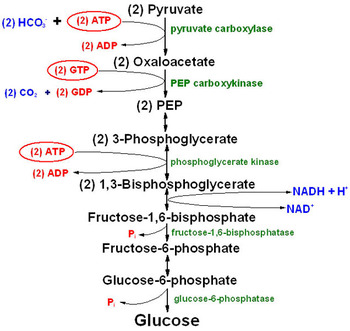
Glucose can be formed from amino acids and from glycerol portion of fat.
Metabolism During Muscular Exercise
Muscle contraction requires much energy, the production of which needs sufficient supply of both O2 and nutrients (glucose). The muscles act as machines for converting the chemical energy into work (Kgm).
Energy Sources For Muscle
The immediate source of energy for muscle contraction is Adenosine triphosphate (ATP) and Creatine phosphate (CP). Creatine is synthesized in the liver from methionine, glycine and arginine in the skeletal muscle.
Creatine is phophorylated to form phosphoryl creatine, which is an important energy store for ATP synthesis, the ATP formed by glycolysis.
During exercise, the reaction is reversed, maintaining the supply of ATP which is the
immediate source of energy for muscle contraction.
Creatine+ATP inverse reaction Phoshoryl Creatine + ADP
However, most of the energy that resynthesizes both ATP and CP is derived from breakdown of glucose or glycogen.
Energy Sources For Muscle
The immediate source of energy for muscle contraction is Adenosine triphosphate (ATP) and Creatine phosphate (CP). Creatine is synthesized in the liver from methionine, glycine and arginine in the skeletal muscle.
Creatine is phophorylated to form phosphoryl creatine, which is an important energy store for ATP synthesis, the ATP formed by glycolysis.
During exercise, the reaction is reversed, maintaining the supply of ATP which is the
immediate source of energy for muscle contraction.
Creatine+ATP inverse reaction Phoshoryl Creatine + ADP
However, most of the energy that resynthesizes both ATP and CP is derived from breakdown of glucose or glycogen.
Aerobic Glycolysis
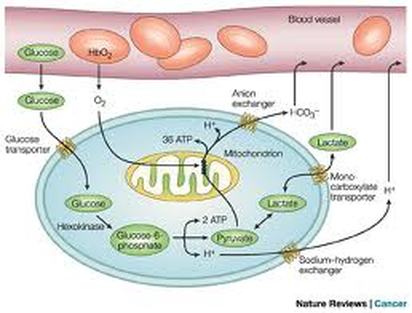
Aerobic glycolysis of glucose to pyruvate, requires 2 ATP to activate the process, with the production of 4 ATP and 2 NADH.
The 2 Pyruvic Acid are converted into 2 Acetyl CoA & this reaction produces 4 hydrogens (2 NADH2).
Those hydrogens (i.e., 2 pair of hydrogens) go through oxidative phosphorylation and produce 6 more ATP.
Overall ATP production from glucose = 8 (from glycolysis) + 6 (from the hydrogens produced when the 2 pyruvic acid are converted into 2 acetyl CoA) + 24 (from the Kreb's cycle).
Overall Total equal to 38 ATP.
The 2 Pyruvic Acid are converted into 2 Acetyl CoA & this reaction produces 4 hydrogens (2 NADH2).
Those hydrogens (i.e., 2 pair of hydrogens) go through oxidative phosphorylation and produce 6 more ATP.
Overall ATP production from glucose = 8 (from glycolysis) + 6 (from the hydrogens produced when the 2 pyruvic acid are converted into 2 acetyl CoA) + 24 (from the Kreb's cycle).
Overall Total equal to 38 ATP.
Anaerobic Glycolysis
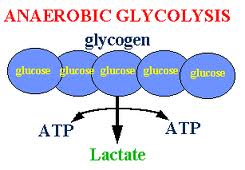
On, oxygen become insufficient, cellular oxidation cannot take palce.
The glycolytic breakdown of glucose to pyruvic
acid do not require oxygen.Pyruvic acid does not enter the Kreb's cycle but is reduced to lactic acid producing little energy as following:
Glucose +2ATP anaerobically 2Lactic acid+4ATP
The glycolytic breakdown of glucose to pyruvic
acid do not require oxygen.Pyruvic acid does not enter the Kreb's cycle but is reduced to lactic acid producing little energy as following:
Glucose +2ATP anaerobically 2Lactic acid+4ATP
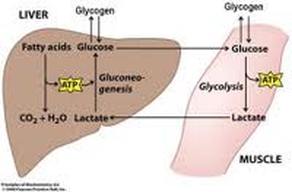
Lactic Acid (Cori Cycle)
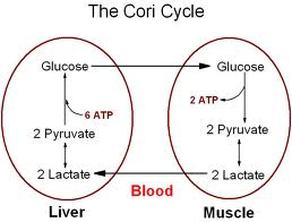
It is pathway by which muscle lactate contributes to blood glucose. Lactate formed in muscle by glycolysis is transported to the liver and resynthesized to glucose there.
Lipid Metabolism
There are three families of lipids:
(1) fats. (2) phospholipids. (3) steroids.

Chemical Composition Of Insect Lipid
The reserve of fat of insects, stored in the fat body, is usually in the form of triglycerides. Analysis of body fats which indicate a high proportion of free fatty acids may be artifacts caused by hydrolysis of glycerides. The fatty acids combined in the triglycerides of insect fats and oils vary in their degree of unsaturation. Although exceptions exist, the fatty acids are usually long chain, and can be saturated acids such as palmitic and stearic acids or unsaturated acids as palitoleic , oleic, linoleic acids. Fatty acids stored as triglyceride can be used for energy production through β-oxidation . Triglyceride is stored in an anhydrous form, whereas glycogen is stored in a bulky hydrated form. Triglycerides also have a higher caloric content per unit of weight than glycogen.
Lipid Stores In Insect
Storage of fatty acids and glucose is essential in insects for other functions as well. Fatty acids serve as precursors in the synthesis of eicosanoids and pheromones, and they are needed in substantial amounts for the synthesis of phospholipids and waxes.
Rediscovery of the Lipid Droplets
Intracellular storage of triglyceride occurs in specialized cytoplasmic compartments called lipid droplets. Almost all tissues can synthesize and store triglyceride in small lipid droplets, but adipocytes are specialized cells for lipid storage. Growing evidence shows that lipid droplets are not just a passive reservoir of lipids but in fact are dynamic organelles serving a central role in fat and energy metabolism.
MOBILIZATION OF LIPID RESERVES
Fatty acids stored in the lipid droplets of the fat body are mobilized for a number of purposes, including the provision of energy to flight muscles, in the form of diglyceride, trehalose, or proline; the provision of lipids to the ovaries; and the overall maintenance of the metabolic activity of other tissues, including the fat body.
The first required step of fatty acid mobilization consists in the action of fat body triglyceride lipases to catalyze the hydrolysis of the triglyceride molecules contained in the lipid droplets. Two lipases that are expressed in the fat body have been identified so far: insect adipose triglyceride lipase (ATGL), or Brummer lipase , and triglyceride lipase (TGL) . Brummer lipase, or insect ATGL, was identified in Drosophila. Insect ATGL belongs to the calcium-independent phospholipase A2 (iPLA2) family, which includes three closely related vertebrate lipases: ATGL, iPLA2-ε, and iPLA2-η.These are phospholipases that also display lipase and transacylase activity.
Utilization of Lipids
This section presents a revision of the studies showing mobilization and/or utilization of lipids for direct support of flight and for the synthesis of trehalose and proline, as well as during starvation, embryogenesis, and the immune response.
Neutral Fat ( Triglyceride )
They are used in the body mainly to provide energy for different metabolic processes.
Triglyceride in formed of three long chain fatty acids bound with one molecule of glycerol:
Triglyceride -------------)- Glycerol + 3Fatty A.
The reserve of fat of insects, stored in the fat body, is usually in the form of triglycerides. Analysis of body fats which indicate a high proportion of free fatty acids may be artifacts caused by hydrolysis of glycerides. The fatty acids combined in the triglycerides of insect fats and oils vary in their degree of unsaturation. Although exceptions exist, the fatty acids are usually long chain, and can be saturated acids such as palmitic and stearic acids or unsaturated acids as palitoleic , oleic, linoleic acids. Fatty acids stored as triglyceride can be used for energy production through β-oxidation . Triglyceride is stored in an anhydrous form, whereas glycogen is stored in a bulky hydrated form. Triglycerides also have a higher caloric content per unit of weight than glycogen.
Lipid Stores In Insect
Storage of fatty acids and glucose is essential in insects for other functions as well. Fatty acids serve as precursors in the synthesis of eicosanoids and pheromones, and they are needed in substantial amounts for the synthesis of phospholipids and waxes.
Rediscovery of the Lipid Droplets
Intracellular storage of triglyceride occurs in specialized cytoplasmic compartments called lipid droplets. Almost all tissues can synthesize and store triglyceride in small lipid droplets, but adipocytes are specialized cells for lipid storage. Growing evidence shows that lipid droplets are not just a passive reservoir of lipids but in fact are dynamic organelles serving a central role in fat and energy metabolism.
MOBILIZATION OF LIPID RESERVES
Fatty acids stored in the lipid droplets of the fat body are mobilized for a number of purposes, including the provision of energy to flight muscles, in the form of diglyceride, trehalose, or proline; the provision of lipids to the ovaries; and the overall maintenance of the metabolic activity of other tissues, including the fat body.
The first required step of fatty acid mobilization consists in the action of fat body triglyceride lipases to catalyze the hydrolysis of the triglyceride molecules contained in the lipid droplets. Two lipases that are expressed in the fat body have been identified so far: insect adipose triglyceride lipase (ATGL), or Brummer lipase , and triglyceride lipase (TGL) . Brummer lipase, or insect ATGL, was identified in Drosophila. Insect ATGL belongs to the calcium-independent phospholipase A2 (iPLA2) family, which includes three closely related vertebrate lipases: ATGL, iPLA2-ε, and iPLA2-η.These are phospholipases that also display lipase and transacylase activity.
Utilization of Lipids
This section presents a revision of the studies showing mobilization and/or utilization of lipids for direct support of flight and for the synthesis of trehalose and proline, as well as during starvation, embryogenesis, and the immune response.
Neutral Fat ( Triglyceride )
They are used in the body mainly to provide energy for different metabolic processes.
Triglyceride in formed of three long chain fatty acids bound with one molecule of glycerol:
Triglyceride -------------)- Glycerol + 3Fatty A.
Triglyceride and Energy
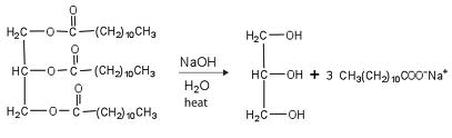
The first stage of triglyceride utilization for energy is hydrolysis of triglyceride into fatty acids and glycerol.
Both products of hydrolysis are transported to the active tissues where they are oxidized to give energy. Almost all cells with except of brain tissue can use fatty acids for energy.
Oxidation of Glycerol
Glycerol is converted to α - glycerol phosphate by glycerophosphokinase
Glycerol glycerophosphokinase α-glycerol-P
ATP --------)- ADP
By NAD-dependent enzyme
α -glycerol phosphate ------------)- dihydroxy acetone-P
Dihydroxy acetone phosphate enters glycolysis to be reduced by enzyme to lactate under anaerobic conditions (1ATP) or to CO2 and H2O under aerobic condition (19 ATP).
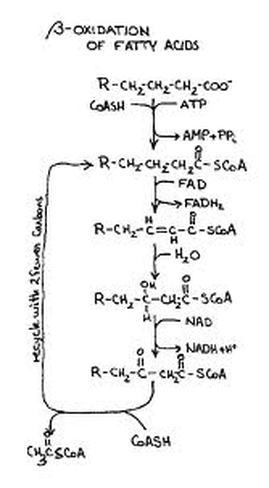
Oxidation of Fatty acids
The degradation and oxidation of fatty acids occur only in the mitochondria. The entrance of fatty acid into mitochondria catalyzed by carnitine enzyme as a carrier substance. Inside mitochondria, the fatty acid splits away from carnitine and is then oxidized degraded into Acetyl CoA fragments.
Oxidation Of Fatty Acid
Both products of hydrolysis are transported to the active tissues where they are oxidized to give energy. Almost all cells with except of brain tissue can use fatty acids for energy.
Oxidation of Glycerol
Glycerol is converted to α - glycerol phosphate by glycerophosphokinase
Glycerol glycerophosphokinase α-glycerol-P
ATP --------)- ADP
By NAD-dependent enzyme
α -glycerol phosphate ------------)- dihydroxy acetone-P
Dihydroxy acetone phosphate enters glycolysis to be reduced by enzyme to lactate under anaerobic conditions (1ATP) or to CO2 and H2O under aerobic condition (19 ATP).
Oxidation of Fatty acids
The degradation and oxidation of fatty acids occur only in the mitochondria. The entrance of fatty acid into mitochondria catalyzed by carnitine enzyme as a carrier substance. Inside mitochondria, the fatty acid splits away from carnitine and is then oxidized degraded into Acetyl CoA fragments.
Oxidation Of Fatty Acid
Protein Metabolism
About 3/4 of the body solids are proteins. These include:
-Structural proteins.
-Nucleoproteins.
-Enzymes and Hormones.
-Protein transporting oxygen.
-Muscle proteins.
Use of Protein in Energy
Excess of amino acids in the body fluids are degraded and used for energy or stored as fat or glycogen. The degradation of amino acids occurs in the liver and it begins with deamination.
Plasma Proteins
Albumin, globulin and fibrinogen are the major types of plasma proteins. Albumin and fibrinogen of plasma proteins as well as 50 % of globulins are formed in the liver the remainder globulin are formed in lymphoid tissues and reticuloendothelial system of cells.The rate of plasma protein formation by the liver reach as much as 100 grams per day.
Functions Of Plasma Proteins
The principle function of the albumin is to provide colloid osmotic pressure and preventing plasma loss from capillaries. The globulins perform a number of natural and acquired immunity against invading organisms. The fibrinogen polymerizes into long fibrin threads during blood coagulation.
Essental and nonessential amino acids
10 of the amino acids normally present in animal proteins can be synthesized in the cells, whereas the other 1o either can not be synthesized. This second group of amino acids that can not be synthesized is called the essential amino acids.
Synthesis of the nonessential amino acids depends mainly on the formation first appropriate α-keto acids that are the precursors of the respective amino acids. Then by process of transamination, an amino radical is transferred to α-keto acid which keto oxygen is transferred of the amino radical.
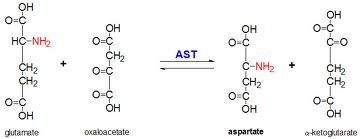
Transamination
The removal of the amino groups of amino acids begins with the transfer of amino groups to just one amino acid - glutamic acid (or glutamate ion). This iscatalysed by transaminase enzymes which transfer the amino group from amino acids to alpha-ketoglutarate. The product is an alpha-keto acid formed from the amino acid and glutamate (formed from the addition of the amino group to alpha-ketoglutarate.
Transamination
-Structural proteins.
-Nucleoproteins.
-Enzymes and Hormones.
-Protein transporting oxygen.
-Muscle proteins.
Use of Protein in Energy
Excess of amino acids in the body fluids are degraded and used for energy or stored as fat or glycogen. The degradation of amino acids occurs in the liver and it begins with deamination.
Plasma Proteins
Albumin, globulin and fibrinogen are the major types of plasma proteins. Albumin and fibrinogen of plasma proteins as well as 50 % of globulins are formed in the liver the remainder globulin are formed in lymphoid tissues and reticuloendothelial system of cells.The rate of plasma protein formation by the liver reach as much as 100 grams per day.
Functions Of Plasma Proteins
The principle function of the albumin is to provide colloid osmotic pressure and preventing plasma loss from capillaries. The globulins perform a number of natural and acquired immunity against invading organisms. The fibrinogen polymerizes into long fibrin threads during blood coagulation.
Essental and nonessential amino acids
10 of the amino acids normally present in animal proteins can be synthesized in the cells, whereas the other 1o either can not be synthesized. This second group of amino acids that can not be synthesized is called the essential amino acids.
Synthesis of the nonessential amino acids depends mainly on the formation first appropriate α-keto acids that are the precursors of the respective amino acids. Then by process of transamination, an amino radical is transferred to α-keto acid which keto oxygen is transferred of the amino radical.
Transamination
The removal of the amino groups of amino acids begins with the transfer of amino groups to just one amino acid - glutamic acid (or glutamate ion). This iscatalysed by transaminase enzymes which transfer the amino group from amino acids to alpha-ketoglutarate. The product is an alpha-keto acid formed from the amino acid and glutamate (formed from the addition of the amino group to alpha-ketoglutarate.
Transamination
Deamination
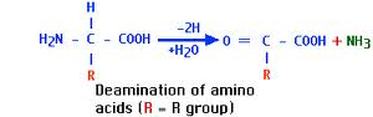
Most of our nitrogenous waste comes from the breakdown of amino acids. This occurs by deamination. Deamination of amino acids results in the production of ammonia (NH3).
Urea Formation
Urea is the chief nitrogenous waste of mammals. Ammonia is an extremely toxic base and its accumulation in the body would quickly be fatal. However, the liver contains a system of carrier molecules and enzymes which quickly converts the ammonia (and carbon dioxide) into urea.
Urea Formation
Urea is the chief nitrogenous waste of mammals. Ammonia is an extremely toxic base and its accumulation in the body would quickly be fatal. However, the liver contains a system of carrier molecules and enzymes which quickly converts the ammonia (and carbon dioxide) into urea.
One Turn Of Cycle
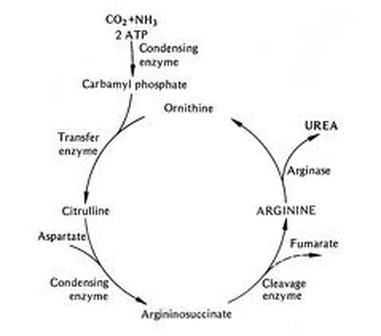
-Consumes 2 molecules of ammonia.
-Consumes 1 molecule of carbon dioxide.
-Creates 1 molecule of urea (NH2)2CO.
-Regenerates a molecule of ornithine for another turn.
-Although our bodies cannot tolerate high concentrations of urea but, it is much less poisonous than ammonia. Urea is removed efficiently by the kidneys.
Failur Of Ammonia Detoxification
There are several inherited diseases of the urea cycle caused by mutations in genes encoding one or another of the necessary enzymes. The most common of these is an inherited deficiency of ornithine transcarbamylase, an enzyme needed for the conversion of ornithine to citrulline. It results in elevated levels of ammonia that may be so high as to be dangerous.
It is an X-linked disorder; therefore most commonly seen in males. It can be cured by a liver transplant. In 1998, an Austrian woman was given a new liver from a male had a mutation in his single ornithine transcarbamylase gene . The woman's blood level of ammonia shot up, and she died a few days later.
-Consumes 1 molecule of carbon dioxide.
-Creates 1 molecule of urea (NH2)2CO.
-Regenerates a molecule of ornithine for another turn.
-Although our bodies cannot tolerate high concentrations of urea but, it is much less poisonous than ammonia. Urea is removed efficiently by the kidneys.
Failur Of Ammonia Detoxification
There are several inherited diseases of the urea cycle caused by mutations in genes encoding one or another of the necessary enzymes. The most common of these is an inherited deficiency of ornithine transcarbamylase, an enzyme needed for the conversion of ornithine to citrulline. It results in elevated levels of ammonia that may be so high as to be dangerous.
It is an X-linked disorder; therefore most commonly seen in males. It can be cured by a liver transplant. In 1998, an Austrian woman was given a new liver from a male had a mutation in his single ornithine transcarbamylase gene . The woman's blood level of ammonia shot up, and she died a few days later.
Uric Acid
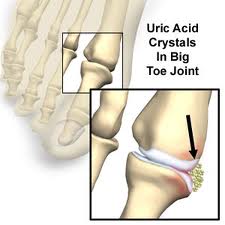
Humans also excrete a second nitrogenous waste, uric acid. It is the product of nucleic acid metabolism. It is produced within peroxisomes. Uric acid is only slightly soluble in water and easily precipitates out of solution forming needlelike crystals of sodium urate. These contribute to the formation of kidney stones. produce the painfull of gout when deposited in the joints.
Uricase Enzyme
Most mammals have an uricase enzyme for breaking uric acid down into a soluble product. However, during the evolution of great monkeys and humans, the gene encoding uricase became inactive.
Uric Acid Crystals
Uricase Enzyme
Most mammals have an uricase enzyme for breaking uric acid down into a soluble product. However, during the evolution of great monkeys and humans, the gene encoding uricase became inactive.
Uric Acid Crystals